|
12/5/2019 Cation Exchange Chromatography
The basic process of chromatography using ion exchange can be represented in 5 steps: eluent loading, sample injection, separation of sample, elution of analyte A, and elution of analyte B, shown and explained below. Elution is the process where the compound of interest is moved through the column. During anion-exchange chromatography for example, negatively charged protein analytes can be competitively displaced by the addition of negatively charged chloride ions (e.g. From sodium chloride). By gradually increasing the salt concentration in the mobile phase, the affinity of interaction between the salt ions and the functional groups.
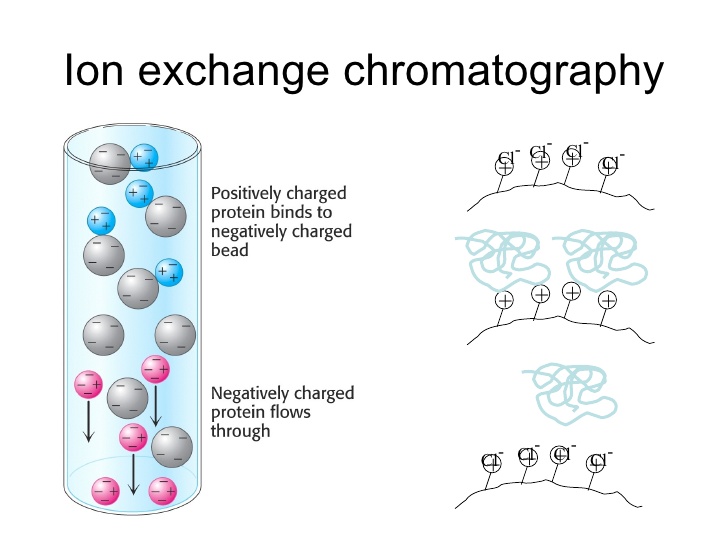 
Anión-exchange chrómatographyis usually a procedure that separates substances structured on their costs making use of an ion-exchange resin filled with positively charged groups, like as diethyl-aminoethyl organizations (DEAE).2In solution, the resin will be coated with positively billed counter-ions (catións). Anion exchange résins will join to negatively charged elements, displacing the countér-ion. Anion éxchange chromatography is usually commonly used to purify proteins, amino acids, sugar/carbohydrates and various other acidic chemicals3with a bad cost at increased pH amounts. The tightness of the binding between the chemical and the resin is usually centered on the strength of the harmful charge of the material.
Common technique for protein purificationedit
A sIurry of resin, like as DEAE-Sephadex is definitely put into the line. The matrix that can be used can be insoluble with billed groupings that are usually covalently attached. These billed groups are referred to as éxchangers like cation ánd anion exchangers. Aftér it forms, the column is certainly pre-equilibrated in buffer before the proteins mixture can be used. DEAE-Sephadex is certainly a positively-chargéd slurry that wiIl have electrostatic connections with the negatively billed atoms, making them elute afterwards than the positively-charged elements in the interested trial. This can be a separation technique utilized widely to find out specific proteins, or nutrients in the entire body.2Unbound proteins are collected in the fIow-through and/ór in subsequent buffer flushes. Protein that content to the positively charged resin are usually maintained and can be eluted in oné of two ways. Very first, the sodium concentration in the elution barrier is slowly increased. The harmful ions in the sodium alternative (at the.g. Cl−) compete with proteins in binding to the resin. Second, the pH of the remedy can become gradually decreased which results in a even more positive cost on the proteins, launching it from thé resin. Both óf these methods can displace the negatively billed protein which can be after that eluted into check pipes fractions with the buffer.456
The separation of proteins will rely on the differences in overall charge. Structure of ionizable part chain groups will figure out the complete cost of the proteins at a particular pH. At the isoelectric stage (pI), the overall cost on the protein is 0 and it will not really hole to the mátrix. If thé pH will be above the pI, the proteins will have a damaging charge and join to the mátrix in an anión exchange line. The stability of the protein at ideals above or beIow the pI, wiIl determine if an anion exchange line or cation exchange column should become utilized. If it will be steady at pH beliefs below the pl, the cation éxchange column be used. If it can be steady at pH values above the pI after that the anion exchange column can be used.7
Personal referencesedit
Gathered from 'https://én.wikipedia.org/w/index.php?name=Anion-exchangechromatographyamp;oIdid=828079088'
Comments are closed.
|
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
||||||||
 RSS Feed
RSS Feed
